 Hence, this is another important difference between oxidation number and charge.Ĭharge and oxidation number are two related terms. The key difference between oxidation number and charge is that we can determine the oxidation number of an atom considering the number of the electron that is either removed or gained by that atom whereas the charge is determined considering the total number of electrons and protons in the atom.įurthermore, there can be several oxidation numbers for the same chemical element depending on the atoms surrounding it while the charge of atom is variable only depending on the number of electrons and protons in the atom. However, there are some differences between oxidation number and charge. Oxidation number and charge are two related terms in chemistry. What is the Difference Between Oxidation Number and Charge? Usually, the same group atoms will form the same charged ions, because they have the same number of valence electrons. We can predict this from the position of the element in the periodic table. Further, the number of electrons donated or abstract differs from atom to atom. There, they gain more electrons than the number of protons resides in their nucleus. Moreover, when an atom is more electronegative, it can attract electrons from other atoms towards itself. Since protons have a positive charge and electrons have a negative charge, when electrons from the valence shell remove, the atom forms a positively charged ion, because then the number of positive protons is higher than the number of electrons in that ion. 
In an atom, the number of protons and electrons are equal. Atoms do removal or gaining of electrons to fill their valence shell according to the octet rule. That is because, the electrons are negatively charged subatomic particles while protons are positively charged. When atoms remove or gain electrons, they obtain an electrical charge. What is Charge?Ĭharge of any atom is zero. For example, if the oxidation number of metal “M” is 3, then we write it as M(III). Moreover, we can denote the oxidation number of the central metal atom with Roman numbers inside brackets. When considering a coordination compound, the central metal atom should always have empty orbitals to which the ligands donate their lone electron pairs and form ionic bonds. Furthermore, transition metal ions (d block) and elements have different oxidation numbers.įigure 01: Determination of Oxidation Numbers of Different Elements in Compounds In a free element, the oxidation number is always zero. 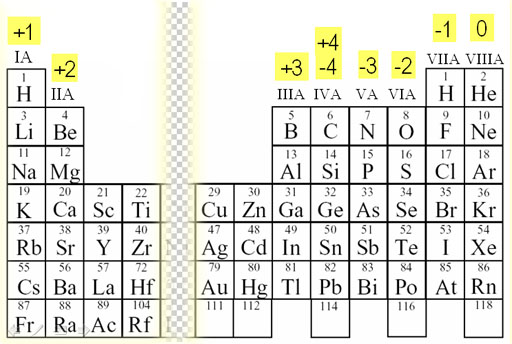
However, the same chemical element can have different oxidation numbers, depending on the other atoms around it. Also, polyatomic ions have the same oxidation number as the charge. For instance, the simple s block and p block elements have the same oxidation number as their charge. 
Therefore, sometimes the charge and the oxidation number are similar, but sometimes it is different. It is the charge of the central atom of a coordination compound when all the bonds around this atom are ionic bonds. Oxidation number is a characteristic of the central atom of a coordination compound. Side by Side Comparison – Oxidation Number vs Charge in Tabular Form Most importantly, it helps to describe the element’s ability to form other molecules and coordination compounds, and thus, helps to identify their empirical formulas. Charge and the oxidation number of an element help to identify, to which group this element belongs in the periodic table. Among a large number of variations between elements, the simplest and important parameters are their charge and oxidation number. And when they join to form molecules, different elements join with other elements in different proportions. The key difference between oxidation number and charge is that we can determine the oxidation number of an atom considering the number of the electron that is either removed or gained by that atom whereas the charge is determined considering the total number of electrons and protons in the atom.ĭifferent elements in the periodic table exhibit different chemical and physical characteristics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
